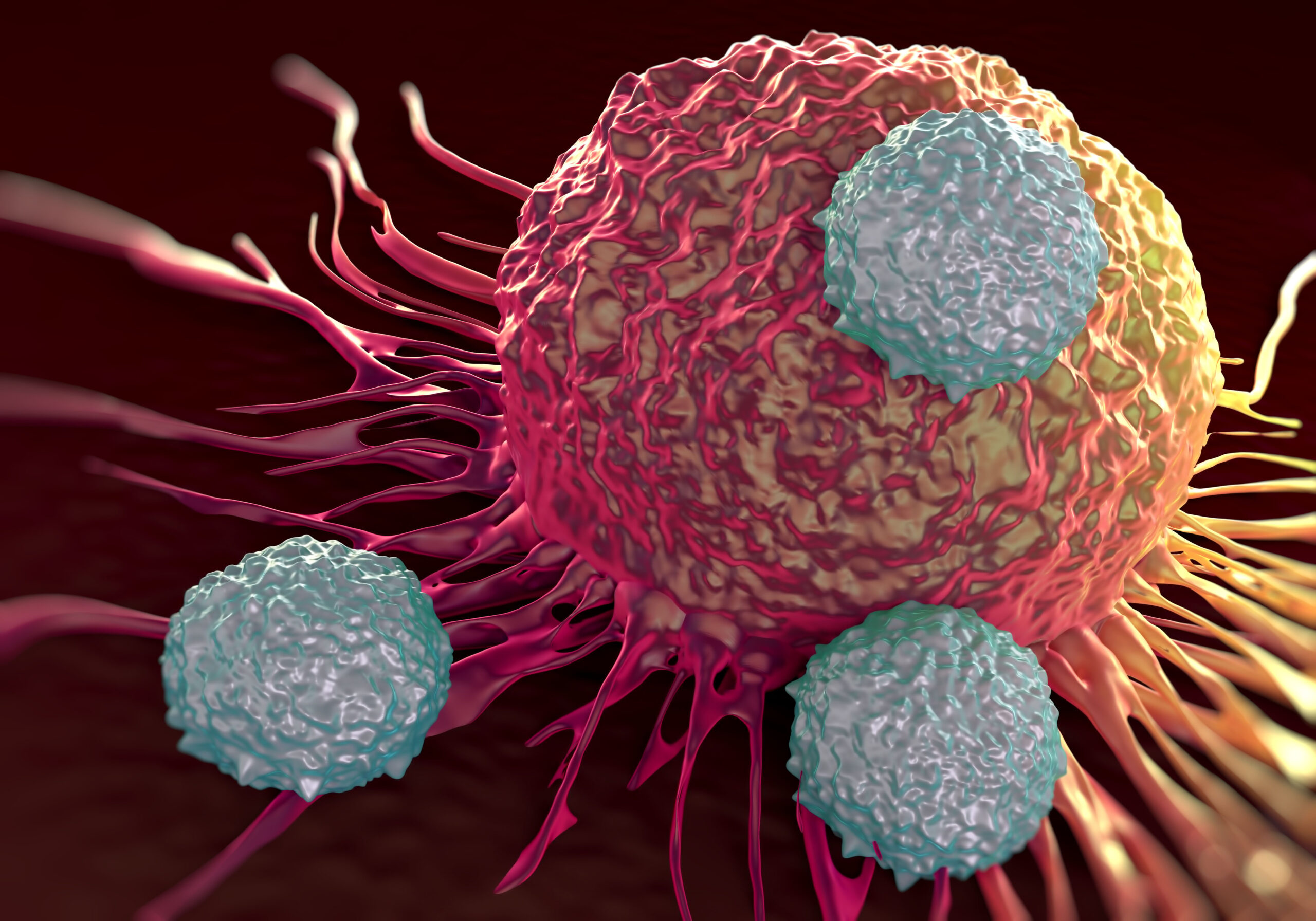
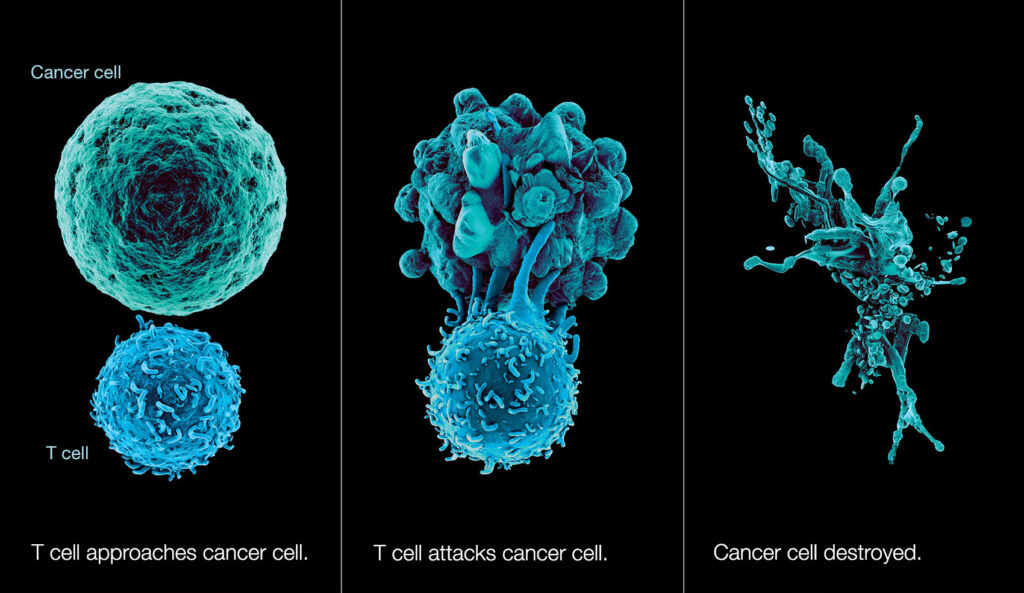
In a groundbreaking paper titled “Naturally occurring T cell mutations enhance engineered T-cell therapies,” scientists from the University of California, San Francisco (UCSF) and Northwestern Medicine have unveiled a revolutionary technique poised to tackle solid tumors. The researchers focused on naturally occurring mutations in malignant T cells causing lymphoma, identified one with exceptional potency, and successfully inserted it into normal human T cells. This innovative approach rendered the normal T cells more than 100 times more potent at eliminating cancer cells without showing any signs of toxicity.
Mutation selected
While current immunotherapies work only against cancers of the blood and bone marrow, the T cells engineered by Northwestern and UCSF turn out to be able to kills tumors developed in the skin, lungs, and stomach of mice and the team is already preparing testing in human.

The key mutation involved a gene fusion, CARD11–PIK3R3, identified in CD4+ cutaneous T-cell lymphoma. This mutation turned out to be able to augment the CARD11–BCL10–MALT1 complex signaling, thus significantly enhancing the anti-tumor efficacy of therapeutic T cells in an antigen-dependent manner in several immunotherapy-refractory models. As observed by the researchers, “Adoptive T-cell therapies have produced exceptional responses in a subset of patients with cancer.”
The safety of this groundbreaking approach was meticulously verified by monitoring CARD11–PIK3R3-expressing cells for up to 418 days after T cell transfer in vivo, revealing no evidence of malignant transformation. These results collectively suggest that harnessing naturally occurring mutations holds promise for deeply exploring T cell biology and for discovering how evolution of malignant T cells can be used as tool to enhance T cell therapies.
Promising approach
Jaehyuk Choi, MD, PhD, an associate professor of dermatology and biochemistry and molecular genetics at Northwestern University Feinberg School of Medicine, emphasizes the transformative potential of their approach: “The superpower that makes cancer cells so strong can be transferred into T-cell therapies to make them powerful enough to eliminate what were once incurable cancers.”
Kole Roybal, PhD, associate professor of microbiology and immunology at UCSF, and center director for the Parker Institute for Cancer Immunotherapy Center at UCSF, adds, “We see this as the starting point. There’s so much to learn from nature about how we can enhance these cells and tailor them to different types of diseases.“
Future projects
The promising results pushed Roybal and Choi to put into practice this approach by starting a collaboration with the Parker Institute for Cancer Immunotherapy and venture capital firm Venrock with the establishment of a new company, Moonlight Bio. The company aims to express the full potential of this technology by developing a new cancer therapy whose testing in humans should begin over the next few years.
This research represents a significant leap forward in the quest for more effective and targeted cancer therapies, offering hope for patients facing previously insurmountable challenges. As scientists continue to explore the intricate landscape of T cell biology, the potential for further breakthroughs in cancer treatment remains on the horizon.